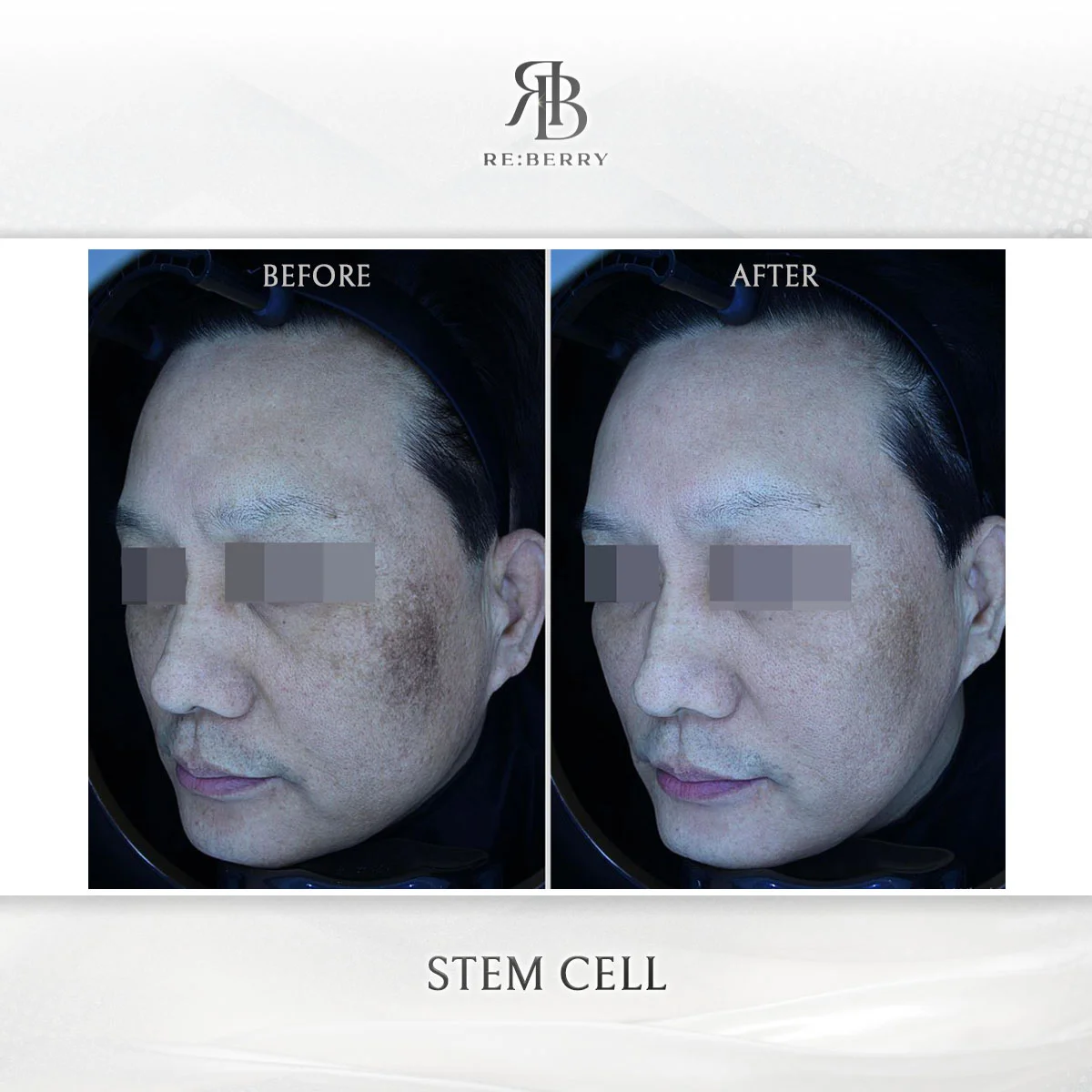
Why Clinic Selection Matters for Stem Cell Therapy
Stem cell and regenerative medicine treatments involve biological materials, specialized processing, and precise delivery techniques. Unlike standard injectable procedures, autologous blood therapy requires centrifugation equipment, sterile processing facilities, and strict cold-chain protocols. The quality gap between clinics directly impacts treatment safety and outcome durability. A poorly processed autologous blood kit loses up to 70% of viable growth factors compared to properly handled samples.
Korea's regenerative aesthetics market has grown rapidly, with the sector expanding 28% annually since 2023. This growth means patients face a wide range of clinic quality levels, making informed selection essential before committing to any treatment program.
Essential Certifications to Verify
Facility and Physician Licensing
Every legitimate stem cell clinic in Korea must hold a KFDA-registered medical facility license. The physician performing the procedure should have a verifiable license number through the Korean Medical Association database, searchable at the Ministry of Health and Welfare website. Confirm the treating physician has completed recognized training in regenerative medicine protocols, as stem cell therapy requires skills beyond standard aesthetic injection technique.
Equipment and Processing Standards
Autologous blood therapy demands medical-grade centrifuges calibrated to separate platelet-rich plasma at 1,200-1,500 RPM for 8-12 minutes. The processing area should be a Class 100 clean room or equivalent laminar flow hood. Ask whether the clinic uses closed-system kits, which reduce contamination risk by 95% compared to open-transfer methods. Facilities using CE-marked or KFDA-approved processing kits provide measurable quality assurance.
Red Flags When Evaluating a Clinic
Be cautious of clinics that refuse to disclose the specific biologics or products used during treatment. Vague descriptions like "stem cell cocktail" or "regenerative blend" without identifying the actual components suggest non-standardized protocols. Clinics that guarantee specific outcomes, such as "permanent rejuvenation" or "complete reversal of aging," misrepresent the realistic scope of regenerative medicine. Current autologous therapies produce results lasting 12-24 months, requiring maintenance sessions.
Price anomalies also signal concern. Autologous blood therapy kits alone cost clinics ₩300,000-₩500,000 per unit at wholesale. A clinic offering complete stem cell packages below ₩800,000 likely uses diluted preparations or uncertified materials. Conversely, pricing above ₩8,000,000 for a single-session protocol without combination treatments warrants scrutiny of what justifies the premium.
Questions to Ask During Your Consultation
Prepare 5 specific questions before your consultation visit. First, ask which autologous processing system the clinic uses and request the brand and model number. Second, inquire about the physician's specific training in regenerative protocols and how many procedures they perform monthly. Third, ask to see the processing area where your biological materials will be handled. Fourth, request documentation of the growth factor concentration achieved by their centrifugation protocol. Fifth, ask about their post-treatment monitoring schedule and what follow-up assessments are included.
A transparent clinic welcomes these questions. Defensive or dismissive responses to reasonable safety inquiries indicate a practice that may not meet the standards required for biological treatment handling.
Comparing Stem Cell Clinics in Gangnam
Gangnam's medical district contains the highest concentration of aesthetic clinics in Korea, with over 450 facilities within a 3-kilometer radius. When comparing stem cell options, create a standardized checklist covering facility certification, physician credentials, processing equipment, published treatment protocols, pricing transparency, and patient follow-up structure. Request at least 2-3 consultations before committing to a treatment plan.
RE:BERRY Gangnam offers autologous blood therapy programs using closed-system processing with Eternal Booster technology. Their regenerative protocols follow standardized KFDA-compliant procedures with documented growth factor concentration benchmarks and structured follow-up timelines at 2 weeks, 1 month, and 3 months post-treatment.